
Introducing Hydrocolloid Wound Dressing Energy Disrupting Human Tissue Metabolism
1 BS MBA,13442 SW 102 Lane Miami, 33186, Florida, United States
|
|
ABSTRACT |
||
|
The
development in 2016 of a novel Potassium Ferricyanide of formula
(K₃[Fe(CN)₆]) based tabletop microscopy method enabled for the
recording of electromagnetic energy emissions from plants and animal tissue.
The microscopy method was subsequently validated and used to document
inter-tissue energy exchanges of both, human blood, and catalase proper with
a hair follicle. As of recent in vitro research using hair follicles as
sentinels support expanding an endogenous irradiation theory as disease
causing mechanism introduced in 1956 and revisited in 2016 to now also
include non-biological exogenous irradiation emitted by hydrocolloid based
wound dressings (WD). Videos and still images are presented validating the
findings of energy emitted by a small fragment of a hydrocolloid based WD
penetrating a 1mm glass slide and unexpectedly delaying the evaporation time
of a Potassium Ferricyanide solution surrounding a freshly plucked human hair
follicle. Absorption of Incoming electromagnetic radiation is a property of
(K₃[Fe(CN)₆]). The introduction of energy from an exogenous
non-biological material, namely a hydrocolloid wound dressing fragment
justifies inclusion in future research protocols. |
|||
|
Received 14 September 2022 Accepted 15 October 2022 Published 31 October 2022 Corresponding Author Abrahám
A. Embí BS, embi21@att.net DOI10.29121/granthaalayah.v10.i10.2022.4836 Funding: This research
received no specific grant from any funding agency in the public, commercial,
or not-for-profit sectors. Copyright: © 2022 The
Author(s). This work is licensed under a Creative Commons
Attribution 4.0 International License. With the
license CC-BY, authors retain the copyright, allowing anyone to download,
reuse, re-print, modify, distribute, and/or copy their contribution. The work
must be properly attributed to its author.
|
|||
|
Keywords: Hydrocolloid Dressing, Exogenous Energy,
Hair Follicle, Potassium Ferricyanide, Human Miniorgan Metabolism, Wound
Healing, Cancer Research Definition of Terms EMFs: Electromagnetic Fields K3Fe: Acronym for the Potassium Ferricyanide formula
K₃[Fe(CN)₆]. SDW: Sandwich. When material is trapped between two
identical glass slides. WD: Wound Dressing. |
|||
1. INTRODUCTION
The
introduction in 2016 of a novel Potassium Ferricyanide (K₃[Fe(CN)₆]) based tabletop microscopy method
enabled for the recording of electromagnetic energy emissions from plants and
animal tissue Scherlag et al. (2016). This microscopy method was subsequently validated
Embi
(2022) and used as tool in introducing biological
inter-tissue energy exchanges from both, human blood, and the ubiquitous enzyme
catalase with a hair follicle Embi
(2020), Embi
(2018). As of
recent in vitro research using hair follicles as sentinels support
expanding the hypothesized intracellular endogenous irradiation theories in
cancer genesis introduced in 1956 and 2016 Harman
(1956), Embi
(2016) to include non-biological xogenous material
irradiation from hydrocolloid based wound dressings (WD). Videos and still
images are presented validating the findings of energy emitted by a small
fragment of a hydrocolloid based WD penetrating a 1mm glass slide and
unexpectedly delaying the evaporation time of a Potassium Ferricyanide crystals
of formula K₃[Fe(CN)₆] in solution surrounding a freshly
plucked human hair follicle. A most
interesting property of K₃[Fe(CN)₆] is the total absorption of incoming
electromagnetic radiation (EMRs) Figgis
et al. (1969), Baranov
et al. (2015); therefore, the presence of an incoming EMR when
encountering K₃[Fe(CN)₆] in solution causes a delay in the
ongoing crystals formation. The changes attributed to incoming radiation in a
human hair follicle placed within the energy cone a WD fragment penetrating a 1
mm glass slide is at the core of this presentation. The addition of energy from
an exogenous non-biological material, namely a hydrocolloid wound dressing
fragment justifies inclusion in future research protocols.
2. MATERIALS AND
METHODS
2.1. Materials
Potassium Ferricyanide powder of formula K₃[Fe(CN)₆].
Two stacked glass slides 25x75x1mm
Bottled water drops
Freshly tweezers plucked human hairs from author.
DuoDerm 1.75 in. x 1.5 in. / 4.4cm x 3.8cm- Hydrocolloid
Wound Dressing cut fragment x4 mm width x 15 mm length. Manufactured by:
ConvaTec Inc. NC
Video-Microscope
MacBook Air Apple computer equipped with photo application
program
2.2. Methods
The hydrocolloid wound dressing (WD) material was cut into
small fragments with a width of approximately 4 mm and 15 mm in length. The
adhesive strip was taped onto a clean 25x75x1mm glass slide. A second like
slide placed covering the WD strip, thus creating a sandwich (SDW). On the top
slide surface of the SDW crystals were placed and diluted by two to three drops
of water, a freshly plucked scalp human hair was aligned with the shadow of the
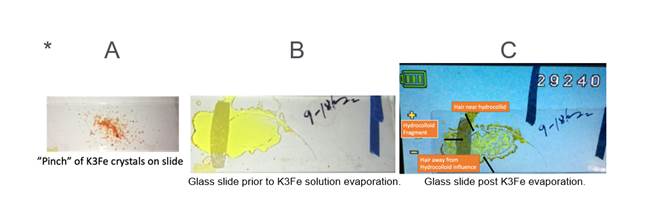
WD. Please refer to (Figure 1), and images A,
B and C below.
Figure 1
|
|
|
Figure 1 Slide Assembly for Experiments |
Hair immersed in
K3Fe solution at 1 mm vertical distance from wound dressing
A narrow
strip of wound dressing (WD) was adhered to a single clean slide (size 25 x 75
x1mm), at this time a second slide was placed on top; thus, creating a sandwich
(SDW). On the top slide of the SDW two drops of K₃ [Fe (CN)₆] were placed, a previously freshly
plucked human hair was situated and maneuvered via toothpick to be near the WD
right shadow’s edge. The diluted K₃ [Fe (CN)₆] crystals and hair are now at a vertical distance
of 1 mm from the WD. The two-slide preparation was allowed to evaporate, still
images and video-recordings taken for further analysis.
Additional
Details
For
simplicity in some areas in the text the acronym K3Fe will substitute the
Potassium Ferricyanide formula K₃[Fe(CN)₆]
On top slide surface dilute a “pinch” * of K₃[Fe(CN)₆] crystals (A below)
in two to three drops of water. Mix crystals with toothpick until in solution,
then place on slide a previously freshly tweezer plucked scalp human hair.
Gently maneuver hair with toothpick until optimal position (B below). Optimal
defined as hair follicle near hydrocolloid shadow now at 1mm vertical distance.
Place B in video-microscope, observe and record changes until C (K3Fe) has
totally evaporated.
TO APPRECIATE DETAILS
 AMPLIFIED
EXHIBIT I POST K3FE EVAPORATION (C ABOVE)
AMPLIFIED
EXHIBIT I POST K3FE EVAPORATION (C ABOVE)
ADDITIONAL NOTE:
The diluted drops on a
slide must be plumb level, this to avoid unwanted liquid shifting on slide. In
the experiments herein presented, notice a left sided placement of the WD; this
to compensate for a slight surface inclination.
3. RESULTS
A total of four in vitro
experiments were done, all detected a disruption of human hairs follicles
electromagnetic radiation as interpreted from the dissapearance of semicircular
concentric K3Fe precipitated crystals circumventing the follicle. Example of a
“normal” image is seen in (Figure 2) below.
CONTROL IMAGE
Figure
2
|
|
|
Figure 2 Control Human
Hair in Liquid K3Fe After Evaporation, Showing: Black
Arrow: Concentric Undisturbed Crystals Around Follicle Indicating Normal Emfs
Waves. Orange Line: Indicating Anatomical end Point Where Follicle’s
Circumventing Emfs are Present. |
EXHIBIT I
Figure 3
|
|
|
Figure 3 Human Hair Away from the
Hydrocolloid Influence at 1 Mm Distance. Blue Arrows: This Time the
Semicircular Concentric EMFs are Documented Absorbed by
the K3Fe. Compare with Figure 2 Above. |
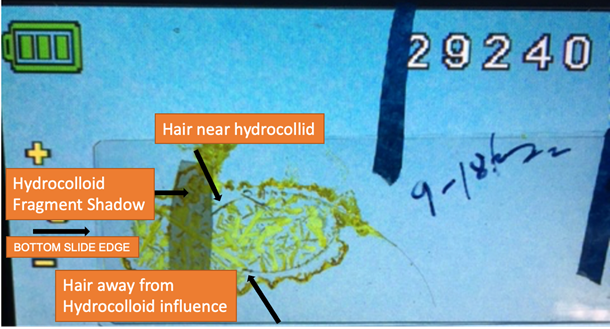
HAIR FOLLICLE NEAR HYDROCOLLOID INFLUENCE SHOWING ABSENCE OF EMF
EMISSIONS.
Figure 4
|
|
|
Figure 4 Hair Follicle
in K3Fe after Evaporation Showing Absence of Semicircular K3Fe Crystals Surrounding
Follicle (See Figure
2, Figure
3). The Absence
is Hypothesized to be from Unexplained to be Named Energy Penetrating the 1
mm Glass Slide from the Wound Dressing Strip. Video frame at 00:04 seconds. |
SAME IMAGE AS Figure 4 SHOWING HAIR
FOLLICLE
Figure 5
|
|
|
Figure 5 Image Showing Effect of Hydrocolloid
Energy on Hair Follicle in K3Fe. For Details, please link to: https://youtu.be/QghUriyCUH4 …..Or Scan QR Code Video frame at 00:39 seconds. |
Figure
6
|
|
|
Figure 6 Another Experiment
Showing Effect of a Sandwiched Hydrocolloid Dressing Strip on Hair Follicle
SSP K3Fe Electromagnetic Emissions. Blue Arrow: Pointing at Aberrant
Potassium Ferricyanide Crystals. Black Arrow: Left Upper Corner Pointing at Shadow’s
Edge of Sandwiched Duoderm Strip. |
4. DISCUSSION
In the text, Figure 2, Figure 3 when compared with Figure 4, Figure 5, Figure 6 show the stark contrast between a “normal” hair
follicle’s energy emission and when the follicle is subjected to a “yet to be
named” energy source emitted by a hydrocolloid dressing fragment. Based on previously published results using a
novel tabletop microscopy published in 2016 relying on a property of K₃[Fe(CN)₆] of total absorption of incoming energy; control images
have documented the human hair follicle as a biological entity emitting energy
(Figure 2). This emission was described as consequence of the
“Inherent electromagnetic forces emanating from human hairs”; subsequent
research using the technique identified the hair follicle as sentinel for drugs
evaluation Embi (2020), Embi (2020) to the transdermal teleportation of hair follicles
energy , up to the interesting
documentation of energy emitted by dead tissue Embi (2022) amongst others.
The hair follicle has been
classified as a miniorgan Schneider et al. (2009) having its own intrinsic metabolism, cell divisions
and an energy emitter. In this manuscript an unknown (yet to be named) energy
source from a hydrocolloid wound dressing is shown to cause noticeable changes
in hair follicles energy emissions. Of interest, is that only when the hair
follicle is placed very near or on a wound dressing strip shadow placed at 1 mm
distance is that the energy emissions are compromised (Exhibit I).
5. Question Arises:
Q: What are the implications of a hydrocolloid wound
dressing energy being transferred onto human tissue?
A: Unknown at present. In this manuscript energy emitted
by a small fragment of a comercially available hydrocolloid dressing (DuoDerm)
is shown to inhibit a human hair follicle emissions of electromagnetic waves as
recorded using a solution of Potassium Ferricyanide (K3Fe). The inhibition was
rationalized by observing a marked decrease in K3Fe crystals surrounding the
follicle. As aforementioned K3Fe absorbs incoming electromagentic radiation.
Further research is warranted.
CONFLICT OF INTERESTS
None.
ACKNOWLEDGMENTS
None.
REFERENCES
Baranov, D. G., Edgar, J. H., Hoffman, T., Bassim, N., and Caldwell, J. D. (2015). Perfect Interferenceless Absorption at Infrared Frequencies by a Van Der Waals Crystal. Physical Review B, 92 (20). https://doi.org/10.1103/PhysRevB.92.201405.
Embi, A. A. (2020). Evidence of Human Inter-Tissue Bioelectromagnetic Transfer : The Human Blood Tissue Intrinsic Bioelectromagnetic Energy Transferring onto a Miniorgan. International Journal of Research -GRANTHAALAYAH, 8(8), 288-296. https://doi.org/10.29121/granthaalayah.v8.i8.2020.1178.
Embi, A. A. (2016). Endogenous Electromagnetic Forces Emissions During Cell Respiration as Additional Factor in Cancer Origin. Cancer Cell Int. https://doi.org/10.1186/s12935-016-0337-y.
Embi, A. A. (2018). Catalase Intrinsic Emissions of Electromagnetic Fields as Probable Cause in Cancerogenesis from Consumption of Red and Processed Meat. International Journal of Research -GRANTHAALAYAH, 6(8), 33-40. https://doi.org/10.29121/granthaalayah.v6.i8.2018.1259.
Embi, A. A. (2020). The Drunken Hair: Introducing in Vivo Demonstration of Increased Blood Alcohol Concentration Temporary Disrupting Human Hair Follicles Emission of Electromagnetic Radiation. International Journal of Research -GRANTHAALAYAH, 8(10), 123-130. https://doi.org/10.29121/granthaalayah.v8.i10.2020.1568.
Embi, A. A. (2020). The Human Hair Follicle as Sentinel for Drugs Evaluation : Demonstration of Tetracycline Adhesion to Hair Follicle as Proposed Mechanism in Dysfunctional Hair Loss. International Journal of Research -GRANTHAALAYAH, 8(11), 324-332. https://doi.org/10.29121/granthaalayah.v8.i11.2020.2521.
Embi, A. A. (2022). Introducing Methodology to Detect Dead Tissue Stored Energy. International Journal of Research - GRANTHAALAYAH, 10(8), 20-29. https://doi.org/10.29121/granthaalayah.v10.i8.2022.4733.
Embi, A. A. (2022). Spontaneous Levitation of Plucked Human Hairs on a Glass Slide When Immersed in Liquid Potassium Ferricyanide. Supporting a Tabletop Microscopy Methodology for the Imaging of Electromagnetic Energy in Plant and Animal Tissue. International Journal of Research -GRANTHAALAYAH, 10(7), 106–111. https://doi.org/10.29121/granthaalayah.v10.i7.2022.4710.
Figgis B. N., Gerloch, M., Mason, R., and Nyholm, R. S. (1969). The Crystallography and Paramagnetic Anisotropy of Potassium Ferricyanideproc. Royal Society, A309(1496), 91–118. http://doi.org/10.1098/rspa.1969.0031.
Harman, D. (1956). Aging : A Theory Based on Free Radical and Radiation Chemistry. Journal of Gerontology, 11(3), 298–300. https://doi.org/10.1093/geronj/11.3.298.
Scherlag, B.J., Sahoo, K., and Embi, A.A. (2016). A Novel and Simplified Method for Imaging the Electromagnetic Energy in Plant and Animal Tissue. Journal of Nanoscience and Nanoengineering, 2(1), 6-9.
Schneider, M.R., Schmidt-Ullrich, R., and Paus, R. (2009). The Hair Follicle as a Dynamic Miniorgan. Current Biology. 19(3), R132-42. https://doi.org/10.1016/j.cub.2008.12.005.
 This work is licensed under a: Creative Commons Attribution 4.0 International License
This work is licensed under a: Creative Commons Attribution 4.0 International License
© Granthaalayah 2014-2022. All Rights Reserved.