|
|
|
|
Original Article
Multi-Omics-Driven Herbal Research: Integrating Metabolomics, Genomics, and Systems Biology to Decode Therapeutic Mechanisms
|
Dr. Kishore
Kumar Godisela 1*, Dr. Parshaveni Balaraju 2 1 Associate Professor of
Biochemistry, Department of Biotechnology Kakatiya Government College,
Autonomous Hanumakonda, Telangana 506001, India 2 Associate Professor of Botany, Government
Degree College Husnabad 505467, Affiliated to
Satavahana University Karimnagar, India |
|
|
|
ABSTRACT |
||
|
Herbal medicines from traditional medical systems function as valuable resources which contain diverse medicinal compounds that can treat multiple medical conditions. The complex multi-component system of these products hinders their ability to meet conventional drug testing standards which focus on single drug and single target methods thus delaying their acceptance as modern clinical treatment methods. Multi-omics technologies which include genomics and transcriptomics and proteomics and metabolomics and systems biology have created a new scientific approach in phytomedicine research. This review examines how multi-omics research methods reveal the complex medicinal functions which herbal bioactive compounds possess. The research shows that genomic and transcriptomic analysis methods enable scientists to understand how phytochemicals control blood sugar levels through their impact on host gene networks and biological processes. The research investigates how the plant metabolomic profile which identifies all its phytochemicals interacts with herbal treatment to show metabolic changes in the host organism. This article demonstrates how systems biology and network pharmacology combine different high-throughput datasets into a unified system. Research scientists create detailed compound-target-disease networks which enable them to visualize how botanical extracts produce combined effects through multiple biological pathways. The development of standardized bioinformatic pipelines and data integration algorithms and the establishment of valid methods to measure the natural biological variability of herbal preparations continue to present major difficulties. The path from multi-omics data collection to the development of practical drug discovery solutions stands as the most important challenge which must be overcome to update herbal medicine practices while creating new network-based treatment methods. Keywords: Multi-Omics, Network Pharmacology,
Systems Biology, Metabolomics, Transcriptomics, Herbal Medicine,
Phytochemicals |
||
INTRODUCTION
Human beings have
historically depended on natural products and herbal medicines as their primary
source of medical knowledge. The research shows that these treatments can
effectively treat chronic and complex medical conditions yet scientists find it
challenging to determine precisely how these treatments work. The composition
of herbal extracts contains more than 100 secondary metabolites which include
alkaloids and flavonoids and terpenoids and saponins that produce combined
effects on multiple biological systems.
Pharmacological
research used a reductionist framework for several decades which sought to
identify one active ingredient that scientists could use to study one
biological target. The method succeeded in producing essential medications such
as artemisinin and paclitaxel but it failed to create a complete picture of how
the entire herbal extract worked together to produce its effects. The research
community has started using a comprehensive top-down method that combines
high-throughput multi-omics with systems biology to solve this research
limitation. This review shows how scientists use genomics transcriptomics and
metabolomics to understand polypharmacological
networks present in herbal medicines.
The Limitations of the Reductionist Paradigm
The standard
process for discovering new drugs has maintained its focus on developing
treatments which follow the "one drug, one target, one disease"
approach. The method develops highly effective special ligands which bind to
distinct proteins through its basis in the traditional lock-and-key system that
governs receptor pharmacology. The method demonstrates success in treating
urgent medical conditions and single-gene diseases but its effectiveness has
started to decline when used for treating complex age-related chronic
conditions and metabolic disorders and neurodegenerative diseases and heart
disease.
The disease
networks which exist in chronic conditions show strong structural resilience
because their design includes multiple pathways and nodes; thus, blocking any
pathway will cause compensatory effects which make single-target medications
lose their effectiveness or produce dangerous side effects. The process of
researchers trying to extract one active phytochemical from a known botanical
medicine leads to the discovery that the extracted substance shows much less
effective clinical results and higher toxicity than the complete plant extract
which proves the shortcomings of reductionist bioassay-guided fractionation.
The Complexity of the Herbal Matrix and the Concept of Synergy
The therapeutic
effectiveness of whole herbal extracts surpasses that of their individual
components because different plant parts interact with each other. Plant
secondary metabolites have developed together to operate as a unified system
instead of existing as separate compounds. The combination of different
elements functioning together creates two different methods of drug action.
·
Pharmacodynamic
Synergy: The extract
contains multiple phytochemicals that interact with different molecular targets
which include receptors and enzymes and ion channels that exist throughout a
common disease network. The herbal matrix produces an intensified therapeutic
impact through its ability to control various disease pathway elements which
surpasses the effects of treating a single pathway element.
·
Pharmacokinetic
Synergy: Certain
constituents within an herbal extract may lack direct therapeutic activity but
play a crucial supporting role by modifying the bioavailability of the primary
active compounds. Supporting phytochemicals have the ability to block
intestinal efflux pumps which include P-glycoprotein and they can also inhibit
the hepatic metabolizing enzymes which include Cytochrome P450. This action
prevents the active therapeutic agents from degrading too early and it improves
their absorption throughout the body.
The Advent of Systems Biology and Multi-Omics
Researchers need
to change their fundamental ways of analyzing data
because they need to study the complex web of interactions between multiple
components and multiple targets. The new analytical framework which systems
biology provides is needed for this particular research work. Systems biology
studies organisms as complete networks which depend on their various biological
parts to work together. Researchers now use high-throughput "omics"
technologies to create maps of biological networks which they study. Genomics
and transcriptomics reveal the genetic predispositions of the host and provide
a comprehensive snapshot of how herbal compounds globally upregulate or
downregulate gene expression. mRNA transcription changes do not produce direct
relationships with changes in physiological functions. The herbal formulation
produces specific metabolic changes which lead to particular physiological
outcomes. Metabolomics functions as the primary method to measure these
physiological outcomes and metabolic transformations which the herbal product
causes.
Rationale and Scope of the Review
The combination of
traditional ethnomedicine with modern bioinformatics establishes a foundation
for the development of evidence-based phytotherapy research. The review
presents a detailed investigation of how multi-omics platforms function in
botanical research studies. The article demonstrates that current experimental
research enables scientists to use metabolomics and genomics together with
systems-level network pharmacology as an effective quantitative method to
unlock the intricate treatment pathways and combined effects and total
medicinal power of herbal remedies.
|
Figure 1 |
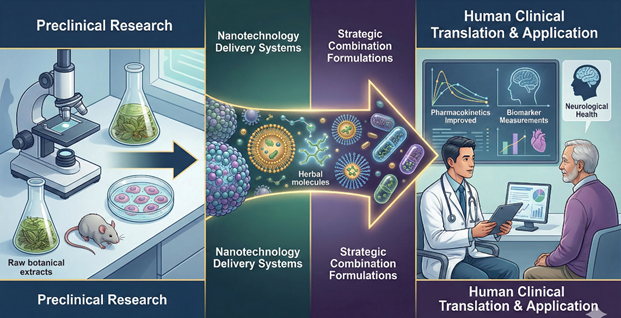
|
Figure 1 Bridging the Gap:
From Lab Bench to Bedside |
The Shift from Reductionism to Systems Biology
Systems biology analyzes biological systems through studying their
molecular interactions instead of examining their single molecular components.
The research shows that herbal remedies work because diseases develop through
multiple genetic changes and protein defects which disrupt entire biological
systems. Herbal medicines work as network therapeutics because they treat
multiple parts of a disrupted disease network to bring back normal body
functions. The researchers need to create complete data sets which show all
genetic and protein and metabolic information about the organism to achieve
their research goals through multi-omics platforms.
The Limitations of the Traditional Reductionist Paradigm
The reductionist
approach which defines modern pharmacological research and drug discovery
through its "one-disease one-target one-drug" model has served as
their primary research method for multiple decades. The "magic
bullet" approach which targets specific diseases through precise treatment
methods has shown success in treating acute and infectious diseases but
encounters difficulties when facing complex chronic metabolic syndrome and
neurodegenerative disorders and cardiovascular disease. Researchers use
reductionism in herbal medicine to study plants by extracting their active
components to examine their effects on specific biological receptors. The
process removes all active chemical compounds found in the entire herbal plant.
Researchers observe that when they test an isolated compound its therapeutic
effects do not match those of the complete botanical extract while the compound
shows unexpected harmful effects which the entire plant does not display.
Systems biology establishes its approach to biological systems by studying all
biological elements because it believes that all physiological networks connect
and complete systems exceed their individual components.
Network Pharmacology and Phytochemical Synergy
The application of
systems biology to botanical research begins with the current development of
"network pharmacology" which represents its most dynamic research
area. The discipline of bioinformatics together with its advanced computational
modeling methods establishes a complete system that
describes all the complicated interactions between multiple bioactive compounds
in herbal treatments and their various human body molecular targets. Herbal
medicines implement their "multi-component, multi-target" approach
because synthetic pharmaceuticals design their products to achieve one specific
target through strong binding power. The various phytochemicals which exist in
an herb show only minor binding strength to particular cellular receptors.
However, when delivered together, they exert a profound synergistic effect. The
combined effects of this synergy enable better treatment results because it
increases the absorption of essential active components while reducing negative
reactions through its controlled impact on multiple linked biological systems.
Multi-Omics Platforms: Decoding System-Wide Responses
Researchers need
to gather empirical evidence which should be collected across complete system
networks to achieve successful mapping and validation of their network
therapeutics. The complete integration of multi-omics technologies establishes
itself as an essential requirement for contemporary herbal research. Scientists
can create a complete multidimensional model which shows how a host responds to
herbal treatment by integrating data from different biological levels:
·
Transcriptomics:
The complete analysis of RNA
transcript data enables researchers to demonstrate how herbal extracts affect
gene expression patterns which result in increased therapeutic pathways and
decreased inflammatory pathways.
·
Proteomics: Because proteins perform essential work to
execute all cellular processes, scientists can study herb-related changes to
cellular functions and signaling pathways and their
results on cell structure by examining proteome transformations.
·
Metabolomics: Metabolomics research tracks the chemical
fingerprints which cells release during their biological activities. The system
analyzes how human metabolism processes plant
phytochemicals and how human metabolism returns to its normal state.
Bridging the Gap with Traditional Knowledge Systems
The transition to
systems biology functions as a crucial link that connects present-day
scientific practices with traditional medical systems including Ayurveda and
Traditional Chinese Medicine (TCM). Holistic principles have guided these
historical frameworks for thousands of years because they view the human body
as an interconnected biological system instead of separate unconnected body
parts. Network pharmacology and multi-omics provide modern science with
analytical tools that enable researchers to decode and quantify traditional
herbal formulations through their complex systemic mechanisms.
Genomics and Transcriptomics: Unveiling Gene Expression Signatures
Understanding how
complex herbal extracts alter the cellular blueprint requires deep molecular
profiling at the nucleic acid level.
Transcriptomic Profiling (RNA-Seq)
High-throughput
RNA sequencing (RNA-Seq) has transformed our
capability to assess the complete time-based changes in host gene expression
that occur after herbal treatment. Researchers use the transcriptomic analysis
of healthy tissues versus diseased tissues and herb-treated tissues to identify
particular pathways that the treatment affects. The transcriptomic analysis of
Salvia miltiorrhiza (Danshen)
extracts shows that the complex formulas which compose the extract downregulate
pro-inflammatory cytokine transcripts while they increase the levels of
antioxidant defense genes in models of cardiovascular
disease.
Epigenomics and Pharmacogenomics
Emerging research
demonstrates that phytochemicals function as epigenetic regulators. The
compounds epigallocatechin gallate (EGCG) and curcumin demonstrate the ability
to change DNA methylation patterns and histone acetylation, which results in
the reactivation of silent tumor suppressor genes.
The study of pharmacogenomics reveals how host genetic variations
(polymorphisms) affect the effectiveness and harmful effects of herbal
treatments, which enables the development of customized phytomedicine treatments.
Metabolomics: The Bridge Between Phenotype and Mechanism
The analysis of
all small-molecule metabolites in a biological system shows the complete
physiological state and physical appearance of an organism. Researchers study
herbal medicine through this method which they use to investigate two different
areas.
Phytometabolomics (Plant Profiling)
The traditional
practice of herbal medicine suffers from its inability to establish proper
quality control methods and standardized procedures. The chemical composition
of a plant changes when it experiences different environmental conditions and
soil types and different times of harvesting. Researchers use
ultra-high-performance liquid chromatography with mass spectrometry and Nuclear
Magnetic Resonance spectroscopy to create precise chemical fingerprints of
herbal extracts. The process guarantees consistency between product batches
while detecting all possible bioactive ligands present in the product.
Host Pharmacometabolomics
The metabolic
balance of the host changes through the administration of an herbal extract.
Researchers can discover endogenous metabolic biomarkers which indicate
treatment effectiveness by analyzing biofluids or
tissues before and after treatment. The treatment responds to multiple herbal
therapies which research shows produce broad effects on metabolic syndrome
through their impact on lipid metabolism and amino acid biosynthesis and
tricarboxylic acid (TCA) cycle functions.
Network Pharmacology: Decoding Multi-Target Mechanisms
The enormous data
output from multi-omics technologies requires advanced computer systems for
proper understanding. Network pharmacology represents the field that connects
systems biology with bioinformatics and polypharmacology
research. The researchers use the TCMSP database and PubChem database to
predict how hundreds of phytometabolites will interact with human protein
targets through computational methods. The system integrates predictions with
established disease-target networks to develop multipartite
"Compound-Target-Disease" models. The topological analysis of these
networks enables researchers to:
·
Identify
Hub Genes: Identify the most
closely interacting proteins such as the targeted protein kinases responsible
for the disease.
·
Predict
Synergistic Pairs: Which
phytochemicals act synergistically to potentiate or diminish the toxic effects?
·
Reveal
Off-Target Effects: Consider conceivable adverse herb-drug interactions
& explain these in terms of categories that are known.
Challenges and Future Directions
While the
multi-omics paradigm offers unprecedented insights, several distinct challenges
must be addressed:
·
Data
Integration: The process of
combining different types of datasets (for example matching transcriptomic
fold-changes with metabolomic flux) presents difficulties in terms of
computational processing. The field needs advanced machine learning algorithms
together with artificial intelligence (AI) technology to achieve effective
integration of multi-omics data into unified biological stories.
·
False
Positives in Network Pharmacology: Many computational target predictions use structural homology and
literature mining which creates a risk of generating incorrect results. The
computational results require verification through in vitro and in vivo testing
which includes CRISPR-Cas9 knockouts & surface plasmon resonance
techniques.
·
Microbiome
Interactions: Future
multi-omics studies must incorporate metagenomics to establish the metabolic
pathways through which the gut microbiome transforms dietary phytochemicals
into biologically active secondary compounds.
Conclusion
The field of
herbal medicine research experiences fundamental change through the application
of genomics together with transcriptomics and metabolomics and systems biology.
The transition to this new approach represents a major shift away from outdated
reductionist research methods which focused on developing drugs that target
specific biological points. Scientists used analytical methods which removed
native phytochemical components from botanical medicines, resulting in their
complete therapeutic potential remaining undiscovered for several decades. The
field of multi-omics-driven network pharmacology now offers an evidence-based
framework which enables researchers to understand the complete synergistic
functions of natural products. Scientists in the current research environment
possess the ability to analyze and comprehend vast
biological data which contains multiple types of biological information. The
development of high-throughput sequencing technologies and advanced
bioinformatics pipelines enables researchers to create detailed models which
show how plant metabolites interact with various human bodily systems. The
development of data integration algorithms together with artificial
intelligence models will improve the speed and accuracy of predicting herbal
product combinations and their possible toxic effects.
The comprehensive
approach which examines entire systems will speed up the process of discovering
new network-based drugs. The future of drug discovery research will focus on
creating multi-component drugs which can restore normal body functions throughout
all disrupted biological systems, instead of finding single active substances
which function as "magic bullets." The present-day world sees this
paradigm shift as a method to unite the two different knowledge systems which
existed between traditional ethnomedical practices and contemporary clinical
methods. Systems biology provides scientific evidence which proves the
effectiveness of ancient holistic healing practices, thus transforming
historical knowledge into validated scientific knowledge. The present-day
convergence between past botanical knowledge and present-day research
establishes a new medical field which enables the safe and effective treatment
of complex chronic diseases using natural products that combine multiple
therapeutic methods.
ACKNOWLEDGMENTS
None.
REFERENCES
Buriani, A., Garcia-Bermejo, M. L., Bosisio, E., Xu, Q., Li, H., Dong, X., ... Williamson, E. M. (2012). Omic Techniques in Systems Biology Approaches to traditional ChinEse Medicine Research: Present and future. Journal of Ethnopharmacology, 140(3), 535-544. https://doi.org/10.1016/j.jep.2012.01.055
Chen, S., Hao, H., Zhao, L., and Wang, G. (2014). Network Pharmacology-Based Drug Discovery and Target Prediction. Current Pharmaceutical Design, 20(15), 2530-2541.
Hao, H., Zheng, X., and Wang, G. (2014). Insights into Botanical Drug Discovery and Development. Nature Reviews Drug Discovery, 13(4), 317. https://doi.org/10.1038/nrd4319
Harvey, A. L., Edrada-Ebel, R., and Quinn, R. J. (2015). The Re-Emergence of Natural Products for Drug Discovery in the Genomics Era. Nature Reviews Drug Discovery, 14(2), 111-129. https://doi.org/10.1038/nrd4510
Hopkins, A. L. (2008). Network Pharmacology: The Next Paradigm in Drug Discovery. Nature Chemical Biology, 4(11), 682-690. https://doi.org/10.1038/nchembio.118
Kellogg, J. J., Kvalheim, O. M., and Cech, N. B. (2020). Developing and Validating Analytical Methods for Botanical Dietary Supplements. Journal of Natural Products, 83(2), 535-551.
Li, S., Zhang, B., and Zhang, N. (2011). Network Target for Screening Synergistic Drug Combinations with Application to Traditional Chinese Medicine. BMC Systems Biology, 5(1), 1-13. https://doi.org/10.1186/1752-0509-5-S1-S10
Nicholson, J. K., Lindon, J. C., and Holmes, E. (1999). 'Metabonomics': Understanding the Metabolic Responses of Living Systems to Pathophysiological Stimuli Via Multivariate Statistical Analysis of Biological NMR Spectroscopic data. Xenobiotica, 29(11), 1181-1189. https://doi.org/10.1080/004982599238047
Pelkonen,
O., Xu, Q., and Fan, T. P. (2014). Why is Research
on Herbal Medicine so Difficult?
British Journal of Pharmacology, 171(19), 4323-4326.
Su, C. H., Lin, Y., and Wang, L. (2021). Multi-Omics Approaches to the Study of Traditional Medicine. Frontiers in Pharmacology, 12, Article 655443.
Ulrich-Merzenich, G., Panek, D., Zeitler, H., Wagner, H., and Vetter, H. (2009). New Perspectives for Synergy Research with the "omic"-technologies. Phytomedicine, 16(6-7), 495-508. https://doi.org/10.1016/j.phymed.2009.04.001
Wang, X., Wang, Z., Zheng, J., and Li, S. (2012). TCM Network Pharmacology: A new Trend Towards Combining Traditional Medicine with Modern Medicine. Evidence-Based Complementary and Alternative Medicine, 2012, Article 789047.
Wishart, D. S. (2016). Emerging Applications of Metabolomics in Drug Discovery and Precision Medicine. Nature Reviews Drug Discovery, 15(7), 473-484. https://doi.org/10.1038/nrd.2016.32
Yang,
Y., Zhang, Z., Li, S., Ye, X., Li, X., and He, K.
(2022). Synergy Between Traditional Medicine and Modern Bioinformatics: Network Pharmacology. Briefings in Bioinformatics,
23(1), Article bbab440.
Zhang, R., Zhu, X., Bai, H., and Ning, K. (2017). Network Pharmacology Databases for Traditional Chinese Medicine: Review and Assessment. Frontiers in Pharmacology, 8, Article 123.
|
|
 This work is licensed under a: Creative Commons Attribution 4.0 International License
This work is licensed under a: Creative Commons Attribution 4.0 International License
© IJETMR 2014-2026. All Rights Reserved.


