|
|
|
|
Original Article
Valorisation of cotton waste for specialised end-use applications
INTRODUCTION
The textile
industry is vast and growing rapidly, but it creates significant pollution
problems due to the waste generated from fabrics made from cotton, linen, and
hemp. This waste originates from the production of clothes, their use, and
subsequent disposal, ultimately filling landfills and releasing harmful gases
while wasting water, energy, and chemicals Chopra et al. (2023).
Fast fashion
enables brands to keep pace with current trends, but it relies on low-cost
materials and rapid production methods, resulting in substantial waste and
environmental pollution. Sustainable solutions like chemical, mechanical, and
biological recycling, along with the concepts of the circular economy, divert
waste, reduce pollution, and promote reuse Nattha et al. (2017).
Strong upcycling
and recycling systems, equipped with cutting-edge sorting and technology,
recover materials for new fabrics or products, thereby reducing the need for
virgin resources Mishra et al. (2022). The recycling of textile waste can be
categorised into either closed-loop or open-loop recycling, with the primary
difference being the product resulting from the recycling process. Closed-loop
cycles reduce the consumption of primary raw materials. Open-loop recycling, on
the other hand, utilises material resources from waste, such as recovered
fabrics and fibres, to create new and different products, often through
processes of downcycling Giuseppe et al. (2025). This research pioneers the sustainable
conversion of post-consumer cotton cellulose, extracted via alkaline pulping
from fabrics, into Nitrocellulose through controlled nitration with mixtures of
nitric and sulfuric acids, substituting hydroxyl groups with nitrate esters to
yield a versatile, flammable derivative.
Nitrocellulose, a
pivotal cellulose derivative, emerges from the nitration of cellulose, a linear
polysaccharide of β-1,4-linked D-glucose units, wherein hydroxyl groups
(OH) at C6, C2, and C3 positions undergo esterification with nitrate groups (ONO₂)
via a mixed nitric-sulfuric acid bath, where sulfuric acid acts as a
dehydrating catalyst to generate Nitronium ions
(NO₂⁺) for electrophilic attack. Producing a highly flammable,
solvent-soluble polymer whose nitrogen content dictates properties: low-N (10.5-11.5%)
forms tough, flexible films soluble in esters/ketones, while high-N variants
(>13%) exhibit explosive detonation velocities up to 7,000 m/s. By utilising
this approach, the research offers a scalable route to circular economy
concepts in materials science while also reducing environmental loads and
opening up applications in lacquers, inks, nail polishes, and smokeless
propellants. To avoid using virgin resources, the current study examines
nitrocellulose synthesis from a variety of cellulosic pre-consumer cotton
scraps, post-consumer textile waste, and wood pulp to identify differences in
purity, nitration efficiency, and resulting stability Sampoompuang et al. (2023).
Materials and Methods
Cellulose-containing
textile waste can arise at various stages of the textile life cycle, including
manufacturing (pre-consumer waste) and consumer use (post-consumer waste).
Pre-consumer waste was collected from Ichalkaranji,
including card noil, comber noil,
and waste sliver. The post-consumer cotton fabric was provided by Bombay
Recycling Concern (BRC). After the hard portion was manually cut and
mechanically sorted, the fabric was shredded in a shredding machine to produce
smaller fibres. After that, any necessary cleaning was finished if required.
Aditya Birla Group provided wood pulp. Nitric Acid 70%, Sulfuric Acid 98%,
Sodium Hydroxide (SD Fine Chemicals Ltd, Mumbai).
|
Figure 1
|
|
Figure 1 Post Consumer
Fabric to Fibre Waste |
Extraction of Cellulose
·
Preparation: waste cotton fibres are sterilised in a 10%
solution of sodium hydroxide (NaOH) at a ratio of 1:20 to separate the
cellulosic pulp. After that, the mixture is heated for three hours at 80°C. The
pulped fibres are filtered out and cleaned with deionised water till the pH
reaches neutral Hasnawati et al. (2020).
·
Drying: waste fibres are preheated to 105°C to
remove moisture and improve the efficiency of replacing the hydroxyl group with
the nitro group.
·
Nitration: conversion of cellulose to nitrocellulose.
·
Cooling: Sulfuric acid and Nitric acid at 20ºC for 10
mins.
·
Adding: 60% Nitric acid into the beaker, and then
slowly add 98% Sulfuric acid. Monitoring: temperature using a thermometer. When
the temperature drops below 20°C, the prepared cellulose pulp is added.
·
Pressing: the cellulose to absorb the solution. Then
treat it with water to neutralise it. After that, press the material as much as
possible and let it dry. When it dries, the colour changes from yellowish to
pale white cotton Khalili et al. (2025).
|
Figure 2
|
|
Figure 2 Cellulose
Nitration in Ice Bath |
Process Optimisation
The changes in
four key parameters (concentration, temperature, duration, and
material-to-liquor ratio) are used to determine the optimal reaction conditions
for the nitration process using waste cotton material. In particular, we intend
to change the material-to-liquid ratio, reaction duration, temperature, and
concentrations of sulphuric acid (H2SO4) and nitric acid (HNO3). We use a
statistical technique called the Box-Behnken method, which enables us to get
the same findings with fewer experimental samples. The Box-Behnken technique is
a response surface methodology that effectively investigates a variety of
experimental factors and their interactions by using a particular design of
trials.
This statistical
technique enables us to significantly reduce the number of trials required
while still producing trustworthy data that help inform the choice of
appropriate reaction conditions for the nitration of waste cotton material. It
minimises resource usage, streamlines the experimental procedure, and lowers
the total amount of time and effort required for the study Mohammed et al. (2021).
|
Table 1 |
|
Table 1 Box-Behnken Method
Response Surface Methodology |
|||||
|
Factor
1 |
Factor
2 |
Factor
3 |
Factor
4 |
Response
1 |
|
|
Run |
Concentration
of HNO3 |
Time |
M.L.R |
Temperature |
Nitrogen
content |
|
% |
Seconds |
ºC |
% |
||
|
1 |
30 |
120 |
15 |
18 |
09.06 |
|
2 |
40 |
180 |
15 |
28 |
10.47 |
|
3 |
40 |
120 |
15 |
23 |
11.01 |
|
4 |
40 |
120 |
20 |
18 |
11.01 |
|
5 |
40 |
60 |
10 |
18 |
11.55 |
|
6 |
30 |
60 |
15 |
23 |
09.07 |
|
7 |
50 |
120 |
10 |
23 |
10.78 |
|
8 |
40 |
120 |
15 |
23 |
09.54 |
|
9 |
30 |
180 |
15 |
23 |
09.21 |
|
10 |
50 |
120 |
15 |
18 |
09.56 |
|
11 |
40 |
120 |
15 |
23 |
10.54 |
|
12 |
40 |
120 |
10 |
18 |
10.09 |
|
13 |
40 |
120 |
15 |
23 |
09.67 |
|
14 |
30 |
120 |
10 |
23 |
9.34 |
|
15 |
40 |
180 |
15 |
18 |
10.08 |
|
16 |
40 |
60 |
15 |
28 |
09.67 |
|
17 |
30 |
120 |
15 |
28 |
09.06 |
|
18 |
30 |
120 |
20 |
23 |
09.14 |
|
19 |
40 |
120 |
15 |
23 |
10.54 |
|
20 |
40 |
180 |
20 |
23 |
10.32 |
|
21 |
50 |
60 |
15 |
23 |
10.01 |
|
22 |
40 |
120 |
20 |
28 |
10.45 |
|
23 |
50 |
120 |
20 |
23 |
10.45 |
|
24 |
40 |
60 |
20 |
23 |
10.11 |
|
25 |
40 |
60 |
15 |
18 |
11.12 |
|
26 |
40 |
120 |
10 |
28 |
10.34 |
|
27 |
50 |
180 |
15 |
23 |
11.01 |
|
28 |
40 |
180 |
10 |
23 |
11.11 |
|
29 |
50 |
120 |
15 |
28 |
09.89 |
Handling and storage of nitrocellulose
It is a highly
flammable material that ignites easily when exposed to low heat. Nitrocellulose
must be handled and stored with care. It is stored in galvanised drums, steel
drums, or cardboard boxes. The storage area must also be maintained dry, cool,
and free of heat sources and flammable objects. An antistatic polypropylene bag
is used to store nitrocellulose.
Results and Discussion
Fourier Transform
Infrared Spectroscopy (FTIR).
Nitrocellulose is
characterised by FTIR spectroscopy, which confirms the degree of nitration and
structural alterations by detecting distinctive absorption bands for nitro
groups. In order to assess the effectiveness and conversion of cellulose to
nitrocellulose component, the presence of chemical functional groups in cotton
waste and nitrocellulose material generated from cotton lint, post-consumer
cotton, and wood pulp was assessed.
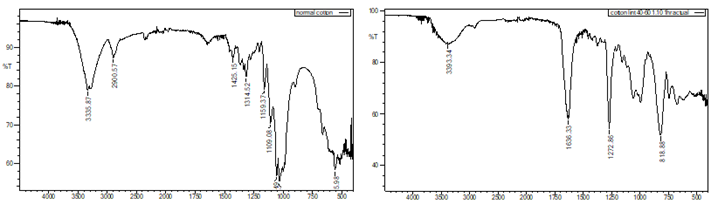
The reaction was
performed on cotton lint, and the FTIR spectra of both the cotton lint and the
resulting nitrocellulose demonstrate similar results. Here are the FTIR graphs
depicting the spectra of cotton lint before the reaction and nitrocellulose derived
from cotton lint. The results are depicted in the Figure below:
|
Figure 3
|
|
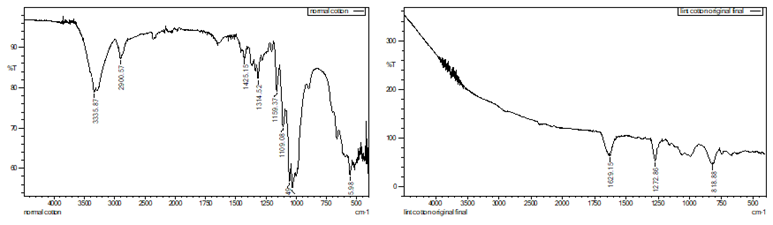
Figure 3 (a) Before
Nitration, (b) After Nitration |
Achieved the best
outcome at the fifth reaction run after optimising the process conditions by
applying Box-Behnken method response surface methodology
|
Figure 4
|
|
Figure 4 (a) Before Nitration, (b) After Nitration |
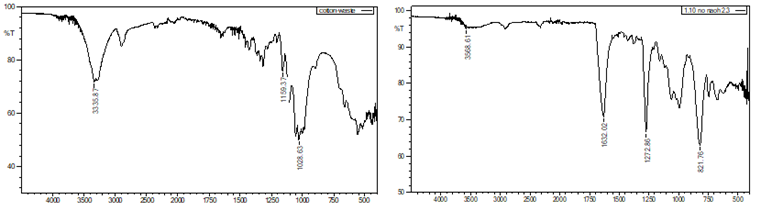
The FTIR spectrum
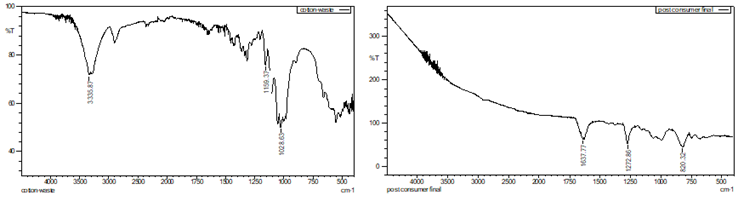
of waste cotton prior to nitration shows the presence of hydroxyl (OH) groups, Figure 4. The stretching vibrations of OH groups are
probably seen as a broad absorption band in the 3200–3600 cm-1 region.
Cellulose, a significant component of cotton, is one of the many organic
molecules that frequently include OH groups. The modified waste cotton's FTIR
spectrum, shown in Figure 4, exhibits a new peak at 1272 cm-1 following
the nitration procedure. The presence of the nitro group (NO2) is shown by this
peak, which is related to the n-o symmetric stretch vibration. The existence of
this peak indicates that nitro groups were added to the waste cotton during the
nitration process.
In the preheating
of cotton fibres at 105°C, extra water will be removed, and the efficiency of
replacing the hydroxyl group by the nitro group may increase.
|
Figure 5
|
|
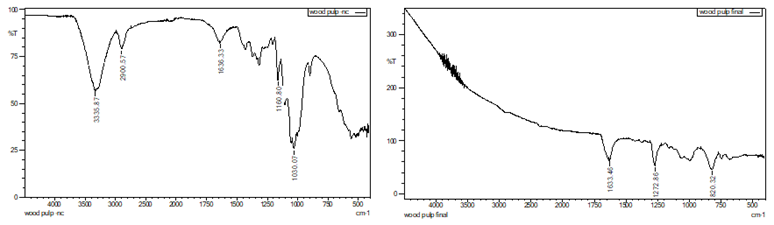
Figure 5 Wood Pulp (a)
Before Nitration, (b) After Nitration |
|
Figure 6
|
|
Figure 6 Pre-Consumer
Cotton Waste (a) Before Nitration, (b) After Nitration |
|
Figure 7
|
|
Figure 7 Post-Consumer
Cotton Waste (a) Before Nitration, (b) After Nitration |
Nitrocellulose
derived from commercial wood pulp, post-consumer cotton waste, and pre-consumer
cotton waste all consistently displayed a strong peak at around 1272 cm⁻¹
in FTIR analysis, which is suggestive of symmetric NO₂ stretching vibrations.
This indicates that nitro (NO₂) functionalities were able to effectively
substitute for hydroxyl (OH) groups in every sample. The optimum pre-nitration
drying temperature for cellulose sources was 105°C, as it eliminates any
residual water that evaporates without breaking the polymer chain. This reduces
hydrolysis side reactions and increases the availability of nitronium
ions for dependable esterification. These results demonstrate the feasibility
of utilising textile wastes as sustainable alternatives to conventional wood
pulp in the synthesis of nitrocellulose, with comparable nitration efficiency.
Carbon, Hydrogen, Nitrogen and Sulphur (CHNS) Elemental Analysis
CHNS elemental
analysis, a combustion-based method that accurately determines the proportion
of Nitrogen (N) together with Carbon (C), Hydrogen (H), and Sulphur (S) by
high-temperature oxidation and thermal conductivity detection, was used to
evaluate the nitrogen content in nitrocellulose samples. Accurate nitro group
substitution levels were determined using this approach, which is crucial for
evaluating the material's viability as a propellant precursor. High-energy
applications are indicated by nitrogen concentrations of more than 12%.
|
Table 2 |
|
Table 2 CHNS Analysis
Results |
||||
|
Sample |
Carbon
(%) |
Hydrogen (%) |
Nitrogen
(%) |
Sulphur (%) |
|
Wood pulp |
28.20 |
4.43 |
11.01 |
Not detected |
|
Pre-consumer
cotton lint |
28.25 |
4.33 |
11.49 |
Not detected |
|
Post-consumer
cotton |
28.25 |
4.45 |
11.17 |
Not detected |
All samples had a
closely clustered carbon content of 28.20–28.25%, which indicates sustained
cellulose backbone integrity after nitrations. Cotton
lint has a lower hydrogen content (4.33-4.45%), indicating a somewhat stronger
nitrate substitution at the C6 hydroxyls. The most obvious difference is in
Nitrogen.
Wood pulp lags
(11.01%), suggesting a 4% relative spread that corresponds to the degree of
substitution, while cotton lint leads at 11.49% and post-consumer cotton is in
the middle (11.17%). To prevent explosive instability or lacquer discoloration,
the consistent absence of sulfur (< detection
limit) verifies clean pulping/nitration.
The
characteristics of post-consumer cotton and cotton lint are nearly identical,
indicating that waste textiles can function as well as virgin lint without
relying on new supplies. The nitrogen content of wood pulp is lower (11.01%),
perhaps as a result of shorter chain lengths or lingering lignin residues.
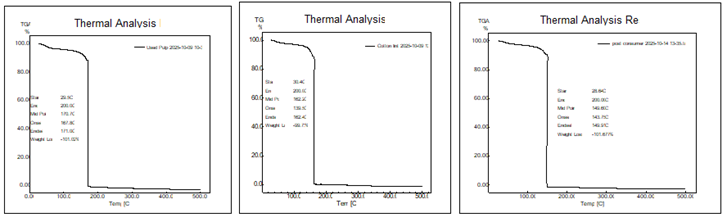
Thermogravimetric Analysis (TGA)
By monitoring mass
loss as temperature rises, thermogravimetric analysis (TGA) assesses
nitrocellulose's thermal stability and breakdown behaviour. Nitrate content
affects nitrocellulose breakdown, as higher nitrogen levels result in lower
decomposition temperatures.
|
Figure 8
|
|
Figure 8 Nitrocellulose
(Wood Pulp, Pre-Consumer and Post-Consumer Cotton) |
|
Table 3 |
|
Table 3 TGA Analysis of
All Samples |
|||||
|
Material |
Onset
(°C) |
Mid
(°C) |
End
(°C) |
Range
(°C) |
Weight
loss (%) |
|
Wood
pulp |
167.84 |
170.78 |
171.04 |
3.20 |
101.023 |
|
Pre-consumer
Cotton |
139.56 |
162.2 |
162.41 |
22.85 |
99.73 |
|
Post-consumer
cotton |
143.75 |
149.60 |
149.91 |
6.16 |
101.67 |
TGA data show that
wood pulp, pre-consumer (cotton lint) and post-consumer cotton, have different
thermal breakdown profiles, emphasising variations in content, purity, and
previous processing that affect thermal stability.
Cotton lint
achieves greater mid (162.20°C) and end points (162.41°C) than post-consumer
cotton (143.75–149.9°C), but it has the lowest onset temperature (139.56°C),
suggesting early hemicellulose or moisture-related breakdown. The shorter
decomposition range of post-consumer cotton suggests that mechanical wear or
contaminants speed its breakdown. Because of its refined cellulose content,
which reduces low-temperature volatiles, wood pulp exhibits exceptional
stability with an onset at 167.84°C and a narrow range (3.2°C span to
171.04°C). All samples approach total mass loss (approximately 100%).
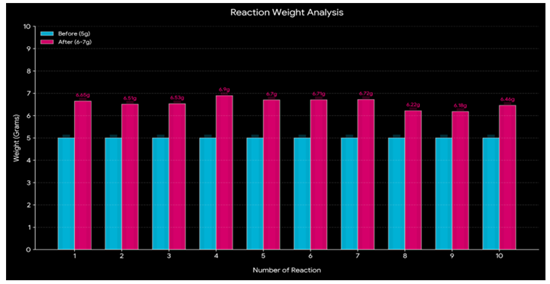
Weight of Cellulose after Nitration
The change in the
weight of cotton waste after the nitration process provides a measure to
estimate the extent of chemical add-on.
|
Figure 9
|
|
Figure 9 Weight Comparison Graph of Cotton Before and
After Nitration |
As seen in Figure 9, the weight of the raw materials, such as
wood pulp and post-consumer cotton waste, including cotton lint fibres,
increases following the nitration process. The incorporation of heavier nitro
groups into the cellulose structure is responsible for this weight shift, which
increases the overall molecular weight.
This is explained
by the reaction's substitution of NO2 groups for OH groups, which raises the
molecular weight. The total atomic mass of the atoms that make up a molecule
determines its molecular weight. When it comes to cotton, the nitro group (NO2)
added during the nitration process has a higher molecular weight than the OH
groups in the cellulose structure. The OH group has a lower molecular weight
(17.01 g/mol) than NO2 (46.01 g/mol). Consequently, the total molecular weight
of the substance rises as the heavier NO2 groups in the cellulose structure
replace the lighter OH groups.
Conclusion
Through optimised
chemical recycling, this study successfully demonstrates the valorisation of
pre- and post-consumer cotton waste, as well as commercial wood pulp, into
high-quality nitrocellulose. Consistent nitration efficiency is demonstrated by
FTIR peaks at 1272 cm⁻¹ (symmetric NO₂ stretch), nitrogen contents
of 11.01-11.49% via CHNS analysis, and comparable thermal stabilities by TGA.
Pre-nitration drying at 105°C emerges as a crucial parameter for optimising
hydroxyl substitution while maintaining cellulose integrity.
A comparative
analysis between nitrocellulose produced by Post-consumer , Pre-consumer cotton
waste and wood pulp reveals pre-consumer cotton lint yielding the highest
nitrogen content (11.49%) indicative of superior nitrate substitution, wood
pulp exhibiting optimal thermal onset stability (167.84°C) due to refined
purity, and post-consumer cotton waste performing intermediately (11.17% N,
143.75°C onset) despite processing impurities yet all samples display
equivalent FTIR signatures and 10-15% weight gain post-nitration, affirming
waste materials as viable sustainable alternatives to virgin pulp. These
discoveries advance the circular economy concepts in materials science by
diverting cellulosic waste from landfills towards the resource-efficient manufacture
of adaptable nitro-esters for lacquers, propellants, and coatings. Future
research may investigate nitrocellulose nanocomposites for high-performance
applications, scale Box-Behnken optimised techniques for pilot production, and
incorporate life-cycle evaluations to quantify environmental benefits, thereby
connecting laboratory innovation with industrial sustainability.
REFERENCES
Chopra, S. S., Liang, D., Kaur, G., Christopher, L., and Lin, C. K. (2023). Sustainable Process Design for Circular Fashion. Current Opinion in Green and Sustainable Chemistry, 39, 100747. https://doi.org/10.1016/j.cogsc.2022.100747
Giuseppe, B., D’Adamo, I., Grosso, C., and Palmieri, R. (2025). Advancing Business Strategy in End-of-Life Management for the Fashion Industry. Business Strategy and the Environment.
Hasnawati, J. S., Roslan, N. J., Shah, N. A., Aminah, S., Ong, K. K., and Wan, W. Z. (2020). Preparation and Characterization of Nitrocellulose from Bacterial Cellulose for Propellant. Materials Today: Proceedings, 29, 185–189. https://doi.org/10.1016/j.matpr.2020.05.540
Khalili, G. A., Ghalehno, M. D., Abadi, S. S., Pouyani, M., and Salimi, A. (2025). A New Process for Producing Commercial Nitrocellulose with Chipping Technology of Sheet Wood Pulp. Scientific Reports, 15(1), 17470. https://doi.org/10.1038/s41598-025-01322-6
Mishra, P. K., Ansari, M., Izrayeel, D., Mathur, B. K., Ahuja, A., and Rastogi, V. K. (2022). A Comprehensive Review on Textile Waste Valorization Techniques and Their Applications. Environmental Science and Pollution Research, 29(4), 65962–65977. https://doi.org/10.1007/s11356-022-22222-6
Mohammed, A., Nasser, M. S., Hussein, I. A., Ahmed, R., and Karami, H. (2021). Application of Response Surface Methodology for the Optimization of the Stability of Fibrous Dispersion used in Drilling and Completion Operations. ACS Omega, 6(4), 2513–2525. https://doi.org/10.1021/acsomega.0c04272
Nattha, P., Leu, S. Y., Hu, Y., and Jing, H. (2017). Recent Trends in Sustainable Textile Waste Recycling Methods. In Chemistry and Chemical Technologies in Waste Valorization (189–228). https://doi.org/10.1007/978-3-319-90653-9_7
Sampoompuang, C., Sukyai, P., Witthayolankowit, K., Lobyam, K., Kampakun, P., Toomtong, R., and Preeyanuch, S. (2023). Recycling of Nanocellulose from Polyester–Cotton Textile Waste for Modification of Film Composites. Polymers, 15, 3324. https://doi.org/10.3390/polym15153324
|
|
 This work is licensed under a: Creative Commons Attribution 4.0 International License
This work is licensed under a: Creative Commons Attribution 4.0 International License
© IJETMR 2014-2026. All Rights Reserved.